Since 1994, the Hong Kong government has been waiving the first registration tax (FRT) for electric vehicles (EV) to encourage the shift from diesel-powered vehicles to EVs and to reduce air pollution caused by the former. According to the Environmental Protection Department (EPD), the number of EVs increased more than a hundredfold from less than 100 in 2010 to 11,018 in late June 2017. However, the number of charging stations lags far behind the vehicles they are supposed to serve. Currently there are 1,505 charging stations in Hong Kong, of which 743 are standard chargers, 509 are medium chargers and 253 are quick chargers. Although the EPD has upgraded 274 chargers from standard to medium in the past three years, progress is yet to match the spike in vehicle numbers.
Increasing the energy capacity in electric car batteries would considerably relieve the burden for charging stations. Scientists have been making efforts in advancing the energy storage technology, especially on the electrochemical front. Redox Flow Battery (RFB) plays an important role in large-scale energy storage applications, especially for electric cars, due to its high security, eco-friendliness, high design flexibility and long life.

Traditional aqueous vanadium redox flow battery (VRB) has a larger power output but a higher cost and lower energy density (25 Wh L-1), which undermines its market competitiveness. In recent years, some forms of semi-solid non-aqueous RFB have shown their potential in boosting energy density, but their power is still lower than the aqueous VRB. In addition, the cost of non-aqueous RFB is significantly higher than that of the aqueous ones.
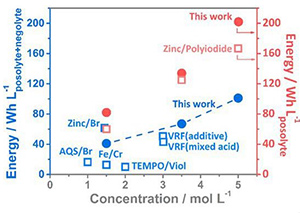
A research team led by the writer has developed a high-energy-density, low-cost zinc/iodine-bromide redox flow battery (ZIBB). It achieves the highest energy density for aqueous redox flow batteries to date via the pioneering introduction of Bromide ions (Br-). The energy density of ZIBB is boosted to as high as 101 Wh L-1, resulting in an improvement of at least 20% in capacity compared to a control system. The breakthrough was published in Energy & Environmental Science in early 2017, and was recently featured in Chemistry World published by The Royal Society of Chemistry.
The Secret of Bromide Ions (Br-): Less Volume yet More Capacity
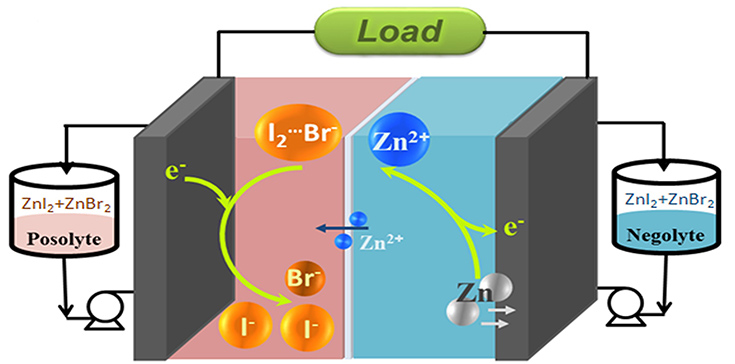
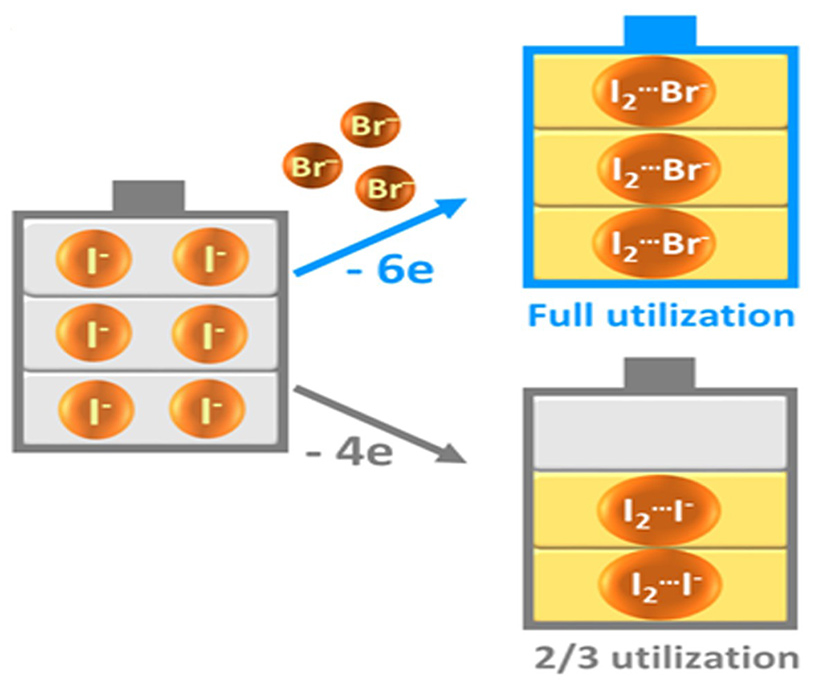
In zinc/iodine RFB, highly soluble zine iodide is the major active material in the electrolyte, with iodide ions (I-) and zinc (Zn) being the electrochemical active ingredients at the positive and negative electrodes, respectively. In the traditional design, the high and stable cycle life (efficiency as high as 95% over 50 cycles) of the battery was ensured by allowing one-third of the iodide ions (I-) acting as a complexing agent to stabilize the iodine (I2), forming triiodide ions (I3-). However, the power of the iodide ions (I-) in contributing to battery capacity is wasted as they are ‘trapped’ as a stabilizing agent.
The team therefore introduced bromide ions as a replacement for the ‘trapped’ iodide ions (I-), i.e., forming iodine bromide ions (I2Br-) by reacting bromide ions (Br-) with iodine (I2). The process still allows for a stable cycle life in the battery, without sacrificing energy capacity.
Expanding energy densities of RFB is considered a substantial contribution to its widespread applications in both stationary and transportation energy storage, especially for large-scale systems. This new flow battery with four times higher energy density than VRF will take up only 0.2 cubic meter to power a 20 kWh household while 1 cubic meter is needed for VRF.
Future Application of ZIBB
This technology has vast commercial potential due to the involvement of low-cost and safe reagents, neutral and mild aqueous electrolytes and low-cost graphite felt electrodes. Both price and charging time of electric cars would be significantly lowered if this type of battery is adopted, with much longer driven mileage. Moreover, the new battery has proved much safer in regular crash tests.
It has also been proved that this innovative concept is also applicable to all electrochemical energy storage systems that involve iodine and is able to improve their theoretical energy density by a third. In the coming few years, the team is going to delve further into the research of low-cost and high-performance electrode and membrane materials to optimize the technology.
The Theme-based Research Scheme (TRS) Project ‘Smart Solar Energy Harvesting, Storage and Utilization’
The battery study is part of the five-year research project ‘Smart Solar Energy Harvesting, Storage and Utilization’ led by Prof. Wong Ching-ping, Dean of Engineering of CUHK. The project has been funded by the Theme-based Research Scheme (TRS) of the Research Grants Council (RGC) of the Hong Kong government (HK$60.33 million) since 2014, with another HK$13.8 million from CUHK and HK$3 million from other partner universities. More than 30 scholars from CUHK, the Hong Kong Polytechnic University, the Hong Kong University of Science and Technology and the University of Hong Kong have been working together to enhance the efficiency of solar power and the penetration of the technology.