Dear readers, With the launch of e-newsletter CUHK in Focus, CUHKUPDates has retired and this site will no longer be updated. To stay abreast of the University’s latest news, please go to https://focus.cuhk.edu.hk. Thank you.
How Do You Mend A Broken Heart?
Kenneth Lee has found a way to creating human tissue

Look through the microscope in the laboratory of CUHK embryologist Prof. Kenneth Lee, and you will see the pumping cells of human heart tissue. That he made.
Professor Lee has worked out how to manufacture human tissue from stem cells, which could lead to the creation of functional patches of tissues for the heart and the liver. These could be used to repair damage to two of the body’s most vital organs.
The heart patches are successful when fabricated in vitro, on a very small scale so far. Professor Lee takes skin cells, then chemically ‘trains’ them to generate pluripotent stem cells, which can become any form of cell in the human body.
But transplanting any such tissue into a human patient is many steps away. Of course, it would take a skilled surgeon. The cells themselves are also a problem. They would need to be pristine, to avoid introducing any infection into a patient. Then there’s the issue that heart cells made outside the body would be contracting at a different rate from the heart in the body.
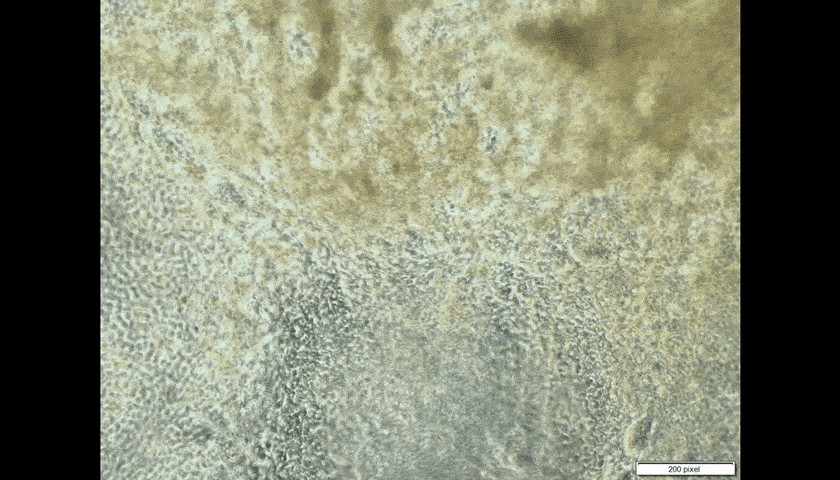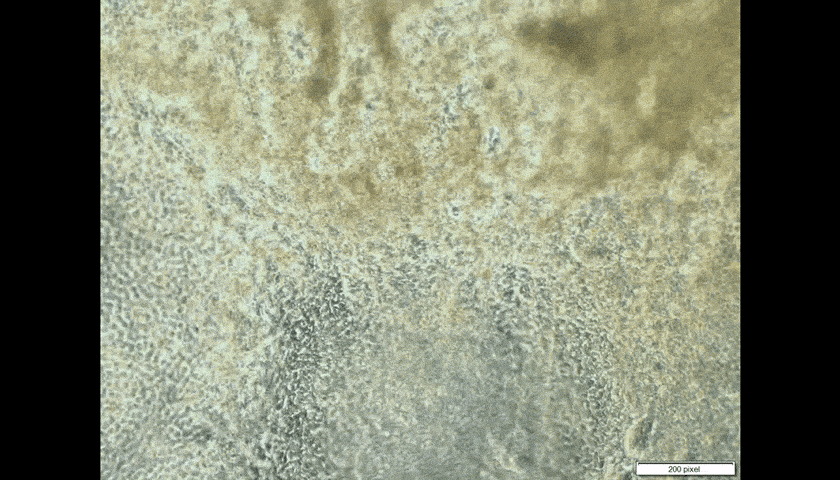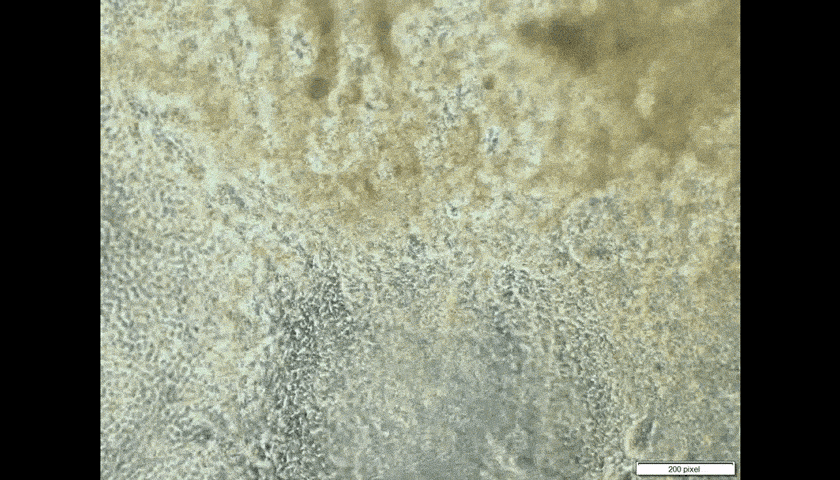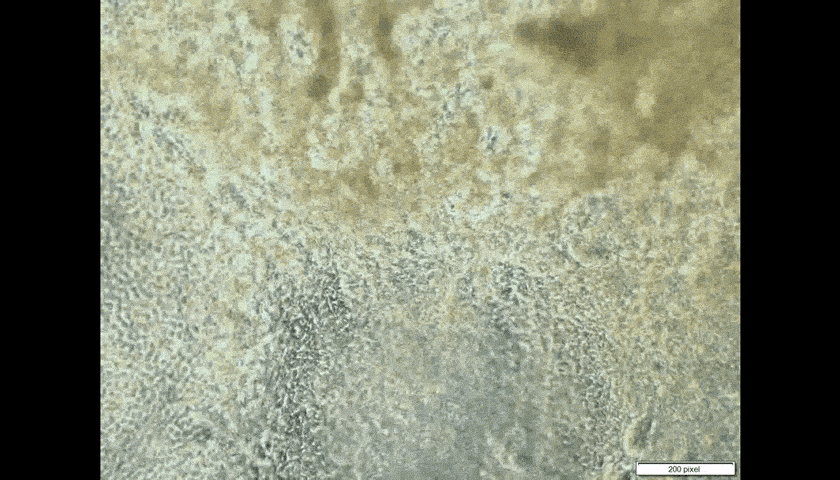
Introducing the heart cells ‘could be counterproductive,’ Professor Lee admits. ‘If they are not properly trained to become heart cells, they could become toxic, could become cancer.’ In the worst-case scenario, pluripotent cells intended to turn into heart cells could morph instead into another type of tissue such as bone.
Professor Lee has formed a company, Stapworks Stem Cell Ltd., that is commercializing his pioneering, patented work. It sells kits that allow researchers and clinicians to produce their own pluripotent stem cells out of fibroblasts, the kind of cells that constitute skin.
Regenerative medicine is a fast-evolving field. Scientists are also looking to repair the human knee, such as a problem joint as people age, by manufacturing cartilage in a petri dish.
The older someone is, the more difficult it is for their own stem cells to replicate. ‘If you can take stem cells from an old person, and replicate them, we have found the rejuvenation elixir,’ Professor Lee says. He laughs at the thought. ‘Hopefully, I can replicate myself.’

The embryologist came to his findings while attempting to recreate research by a Japanese scientist who claimed to have turned skin cells into stem cells using an acid bath. Professor Lee actually debunked this fraudulent claim, and the research was retracted. But he saw a very low level of expression of stem-cell genes being expressed in one of his experiments. He had a hunch he might be able to encourage the expression of stem-cell genes with certain small molecules.
His hunch proved correct, and he was able to encourage the expression of three genes associated with the creation of pluripotent stem cells: Oct4, Sox2 and Nanog. ‘When those genes are expressed, you know they’re stem cells,’ he explains.
Pluripotent stem cells are like troubled teenagers: they’re able to grow to become virtually anything, but are unruly. They need coaxing to become the right kind of cell. Researchers often therefore prefer to work with adult stem cells, which are multipotent, restricted in their ability to become only, say, bone, or muscle tissue, although with a range of possibilities within that category.
Adult stem cells can only replicate for around 50 generations, however, at which point they enter cellular senescence. Pluripotent cells can replicate essentially forever. One kit from Stapworks, in other words, can go a long way in the lab.
Professor Lee, who is a professor in CUHK’s School of Biomedical Sciences, is also working with CUHK’s Department of Surgery in a bid to use stem cells to form soft cartilage that could be shaped into ears. The aim is to replace the tiny nub of an ear that around 20 small children in Hong Kong have at birth, thanks to a condition known as microtia. That might stand an earlier chance of success, since ear cells could be built up and shaped using a 3D bio-printer, then essentially grafted onto the outside of the child’s body.
For now, there is plenty of work developing the basic science of regenerative medicine. Professor Lee’s work is in the phase of developing prototypes, and then taking those into a proof of concept. Beyond that, the applications of man-made tissue are many, and promising indeed.
By Alex Frew McMillan
This article was originally published on CUHK Homepage in Jun 2019.