Dear readers, With the launch of e-newsletter CUHK in Focus, CUHKUPDates has retired and this site will no longer be updated. To stay abreast of the University’s latest news, please go to https://focus.cuhk.edu.hk. Thank you.
Unlocking the difference between the sexes in liver cancer
Why men contract liver cancer far more often than women

The difference between the sexes in contracting various diseases is what fascinates Prof. Alfred Cheng Sze-lok. After first studying estrogen and its impact on breast cancer, he has shifted focus to analyze how the male sex hormone connects to a high incidence of liver cancer.
In men, liver cancer is the second-most common cause of cancer deaths worldwide. It also has a poor range of treatments. The disease is far more prevalent among men than it is with women. The ratio of incidence ranges from 2:1 to as much as 5:1 in some nations.
‘Why are men at a loss?' Professor Cheng has puzzled over what he calls a ‘remarkable phenomenon.'
The first response is to blame the ‘bad habits' that men tend to pursue more than women: drinking and smoking. Obesity and hepatitis viruses also play a part. But experiments in animals have shown those extrinsic factors are not enough to explain the gender gap. When exposed to the same ‘insult,' such as a hepatitis virus, male mice still have a much-higher likelihood of contracting liver cancer than female ones.
Androgen is the male sex hormone that governs the appearance, muscle tone and strength of males. If it is overstimulated—perhaps by the Hepatitis B virus that is most common in China, or the Hepatitis C found very often in Japan—it can lead to the development of cancer. Being overweight can also release the same effect.
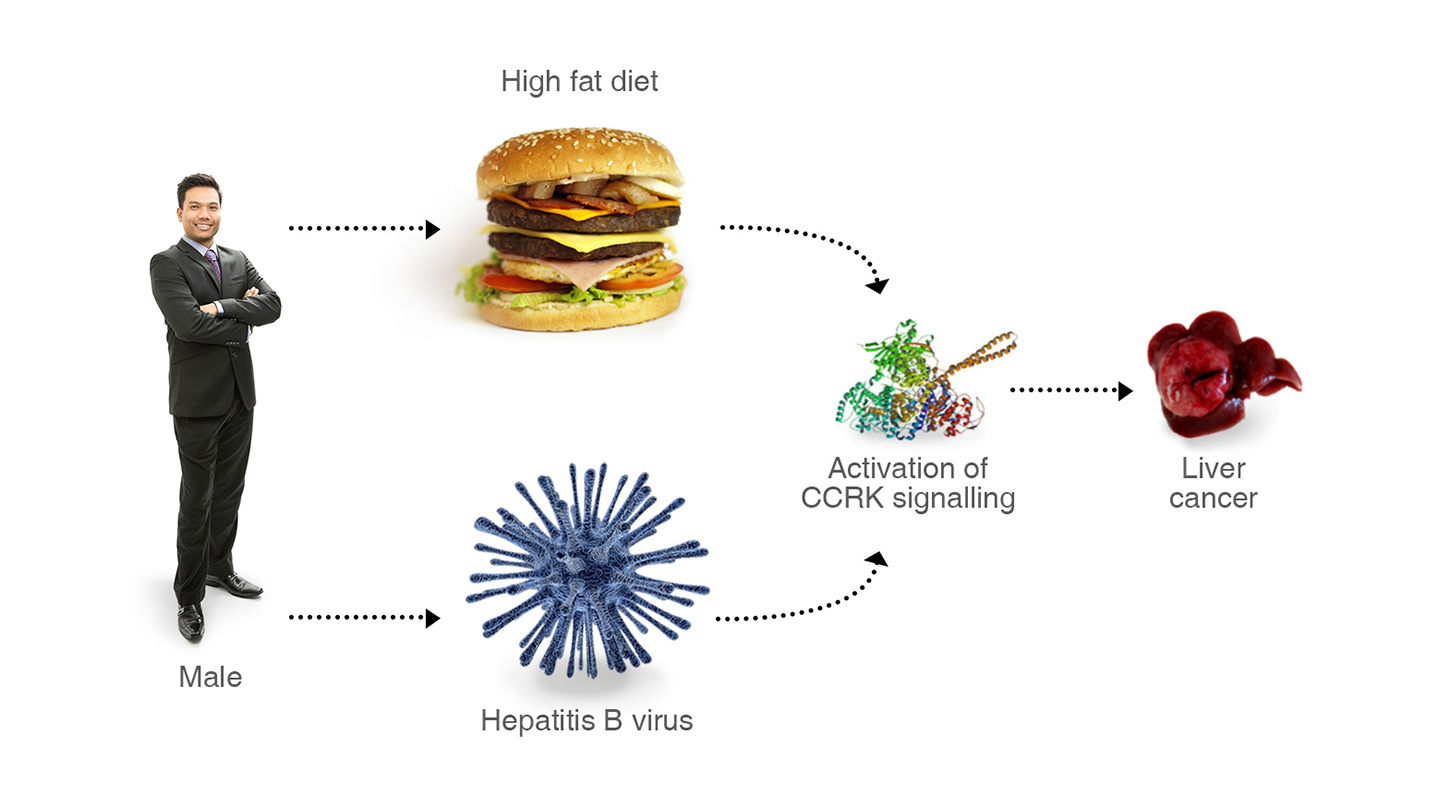
His research has honed in on the molecular activity promoted by the male sex hormone in androgen-receptor pathways. If the receptor is overstimulated, it interacts with DNA to activate a cancer-promoting gene, cell-cycle-related kinase (CCRK), that is a potential carcinogen. Professor Cheng has found that CCRK is silent in a normal liver. But in a diseased liver, it can produce abnormal cells, and ultimately tumors.
No one had identified CCRK before, and it’s exacting work to pinpoint such a gene among the estimated 25,000 in the human body. Of the 200 human samples examined together with clinicians, two-thirds had an over-activated CCRK pathway.
'The more we study this gene the more different functions we find,' Professor Cheng notes. No one is yet sure what function if any it serves in a healthy liver. But highly expressed CCRK in a diseased liver modifies proteins that stimulate key 'signalling cascades,' ultimately leading to liver cancer.
Even worse, it appears to suppress the immune system of cancer patients, according to the latest findings from Professor Cheng’s team. 'CCRK can alter or affect the phenotypes of one type of immune cells, and change them from helping the body to being harmful to the body,' he explains. So CCRK poses two threats to men.
Combatting the disease is vital. Most liver-cancer patients remain asymptomatic and undiagnosed until they have an advanced stage of the disease. At that point, it resists most conventional chemotherapy treatments. The survival rate beyond five years is only 18%. The U.S. Food and Drug Administration has approved only one drug, sorafenib, for late-stage liver cancer. It normally only extends life by a couple of months.
Professor Cheng, who holds a post in CUHK’s School of Biomedical Sciences, came to his key findings via a genome-wide analysis of more than 17,000 genes. He found that around 250 genes bind with the androgen receptor. Computational software drawing on the known functions of those genes then identified around 20 that are particularly influential in cell growth. He then pinpointed CCRK as the likeliest to study in cancer formation because this gene binds the most actively with the androgen receptor.
Cross-studies in cellular and animal models validated the approach. CCRK acts as an important 'signalling hub' within a gene network. His team also found that CCRK may be induced by obesity and diabetes, twin epidemics worldwide. These findings recently lured sizeable funding from the Hong Kong government to conduct a more-detailed investigation.
'As the research goes on, we’re finding more and more interesting things,' Professor Cheng says. 'Kinase is a very promising target for cancer treatment, and it’s important to focus as a researcher or scientist.'
Ultimately this should lead to implications for clinical medicine, including the production of novel molecular therapeutics to reduce liver-cancer risk. Professor Cheng has been working with drug-discovery experts at the University of Sussex to develop small molecules that inhibit CCRK. He also stays in touch with clinical doctors at the Prince of Wales Hospital to keep tabs on what they are seeing first-hand.
'This complimentary strength and mutual interest is very important,' Professor Cheng notes. Although he is now a molecular biologist, he draws on his background in clinical work during his doctorate.

Professor Cheng originally studied how estrogen regulates breast cancer while pursuing that at Ohio State University. At CUHK, he was encouraged by Vice-Chancellor Joseph Sung to look at liver cancer in males, since it is prevalent in Hong Kong.
The drug-discovery process now involves chemists, computer engineers and oncologists around the world. Professor Cheng would like to work with even more collaborators, to form a network that can work towards generating a drug for clinical use. His findings have appeared in noteworthy articles in publications such as the Journal of Hepatology and the Journal of Clinical Investigation.
While he calls that cross-disciplinary approach exciting, it is equally challenging. The drug-discovery process requires a cross-border effort that may well take years to bear fruit. At this stage, it is not yet clear that it will, although chances look promising. Even if the work succeeds, drug approval takes virtually a decade to secure.
'I really hope that CCRK targeted therapy can be actuated,' Professor Cheng says. 'But for the drug development, we still have a long way to go.'
By Alex Frew McMillan
This article was originally published on CUHK Homepage in Aug 2016.