Dear readers, With the launch of e-newsletter CUHK in Focus, CUHKUPDates has retired and this site will no longer be updated. To stay abreast of the University’s latest news, please go to https://focus.cuhk.edu.hk. Thank you.
The Angel’s in the Details
Zhou Renjie captures the infinitesimal with his super microscope

Nothing spells out ‘Science Lab’ better than the image of a microscope. Together with the white lab coat, these are the stereotypical tools of a scientist’s trade.
But microscopes have come a long way since the days of our high school biology class. At CUHK, Prof. Zhou Renjie has invented a 3D light microscope that is so quick, precise and higher in resolution that it can image the movements of individual cells in real time, in three dimensions.
The new device, which he calls the High Speed Tomographic Phase Microscope, is so fast and powerful that it can image the dynamics of cellular compartments such as membranes or nuclei, how sperm cells move, how bacteria change structure or ‘swim’ around a cell body. It can benefit biologists who need to visualize cell structures, just as well as material scientists who want to inspect the structures of various materials at a microscopic level.
‘This new microscope invention can potentially revolutionize traditional biological imaging,’ Professor Zhou says. He now directs CUHK’s Laser Metrology and Biomedicine Laboratory, which works on developing new imaging techniques for solving complex problems in biomedicine and material science.
The new microscope can be used to deliver blood tests and screening results within minutes that would normally take days. We are used to having blood drawn, then waiting while it’s sent to a lab and processed, only for us to have to make an appointment on another date to hear the results. What if the whole process of blood testing or tumour detection could be achieved virtually on the spot, in real time?

Another great advantage of Professor Zhou’s method of processing cells is that it is ‘label-free’. Current cell analysis techniques require chemically staining the cells to make them visible. More than likely, the cells also need to be dead, either so they are conductive or because they are killed by the chemicals themselves.
The new microscope can analyse living cells in their native state, in real time. It is so fast that it can perform observations at more than 600 tomograms, or three-dimensional cross sections, per second. That translates into capturing one 3D cell image at less than 1.7 thousandths of a second, at least 10 times faster than existing 3D cell imaging techniques.
Professor Zhou, who is an assistant professor in the University’s Department of Biomedical Engineering, began investigating this field as a doctoral student, inventing a 3D cell-imaging technique while completing his PhD at the University of Illinois. But he was held back by the ability of camera image sensors to capture images at the appropriate level of detail, the quality of mechanical scanners to manipulate laser beams, and the power of software to process the images.
The new microscope is so detailed that it requires intense computing power to process the information. The capabilities of the camera hardware, mechanical scanner and computer software began to catch up to Professor Zhou’s vision three years ago. That’s when he built the first version of this system, which could image at 30 tomograms per second.
Professor Zhou’s system manipulates light at a microscopic level. Millions of tiny mirrors, only a few microns in size, are driven by an electrical current to move incredibly fast, some 10,000 times per second. This array of fast-moving mirrors acts like a superhigh-resolution projector.
This enables the analysis of how the organelles that are parts within cells move, with some cell structures changing incredibly rapidly. In the past, those were viewed in two dimensions, or in three dimensions but as static images. Now, the new microscope can ‘watch’ those cell parts in three dimensions, in real time.
As well as very fine detail, the microscope can also assess cells in great numbers. In a few minutes, it can image a few million cells. Since a tumour cell, for instance, may be only one part in one million in blood, this scope of detail could allow for early detection of rare cancers.
It’s also possible, since the cells don’t need to be dyed or treated with chemicals in any way, for laymen who are not doctors or biologists to take cell images, and analyse them.
Since designing the system in 2018, Professor Zhou and his team have made the microscope 10 times faster. They have also been optimizing the image-processing software to image cells in even greater detail, while avoiding blurring.
Fine-tuning the microscope precisely so that light moves through the whole optical system correctly takes skill learnt through hours in the lab. ‘If you do it so many times, you have experience on how to make it faster and more precise,’ Professor Zhou says. ‘There are some tricks that you learn over the years. Now I can align a laser beam to go through a 10-micron pinhole, or one thousandth of a millimetre, within a few minutes.’
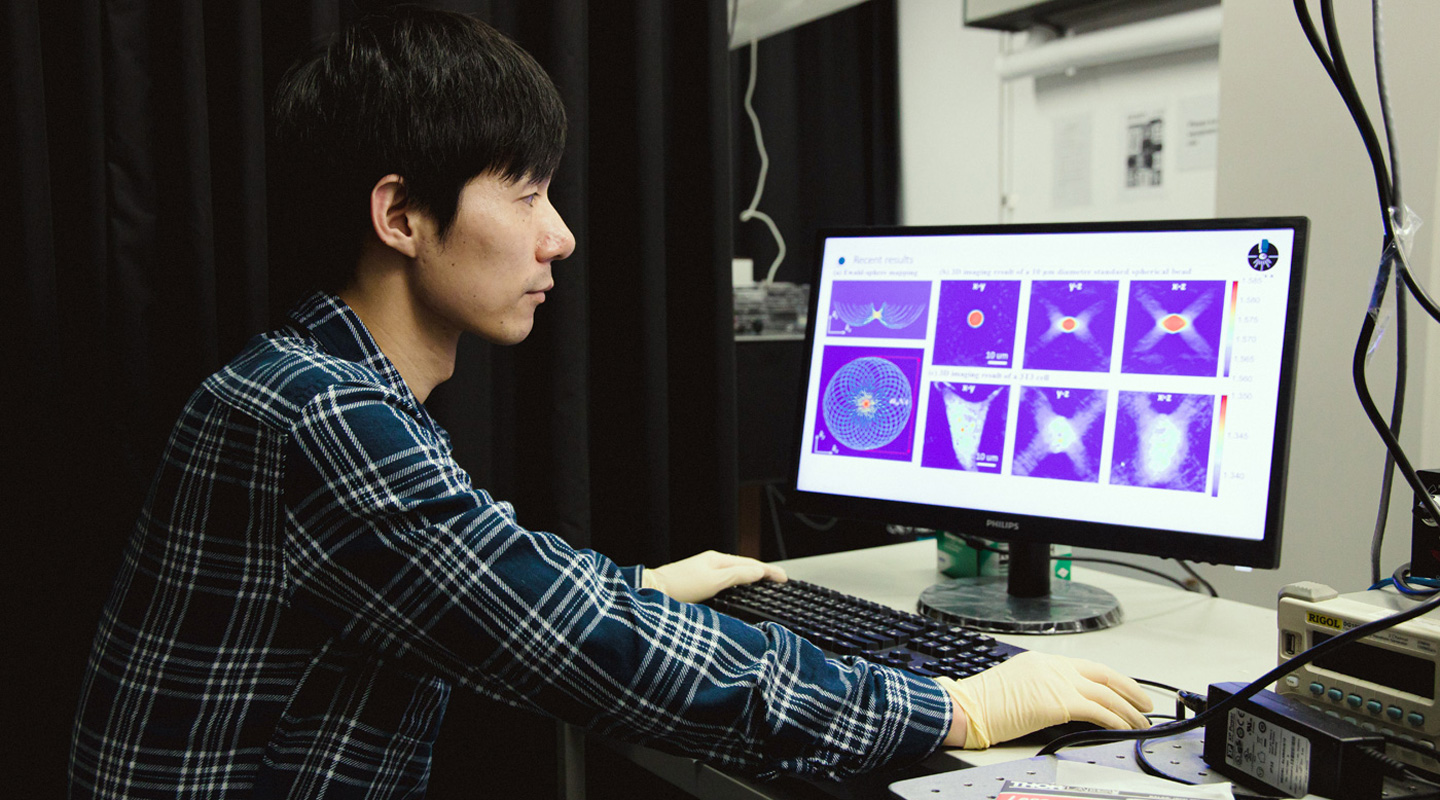
While in the popular imagination, new software draws all the headlines, Professor Zhou’s hardware is in high demand for collaborations with scientists. He is working with biologists studying how the tails of sperm move, another group who want to see how drugs are delivered into cells, yet another team working on mechanically stretching cells. He is also working with a neurosurgeon on a proof of concept in detecting the severity of tumours in brain tissue.
‘The doctor is very excited about this, and has proposed to try it in clinics,’ Professor Zhou says. ‘The microscope is so fast, doctors can do pathology during an operation. A biopsy takes hours to examine. Now doctors can look at it in real time, and can tell you whether there’s a tumour margin or not. They don’t want any small tissue of the tumour remaining, or the patient needs to do the operation again.’
These collaborations are necessary, and excite him, since they put his invention to work. ‘Everyone is doing the software – not many people do the hardware,’ Professor Zhou says, comparing his lab to a hardware store. ‘I think of my lab as The Home Depot, where people go to buy tools. We provide this instrument, this tool for biologists, doctors, material scientists. How good it is is based on their usage, and whether they have solved their problems.’
By Alex Frew McMillan
Photos by Eric Sin
This article was originally published on CUHK Homepage in Mar 2020.